Why there is different between the value of compressibility factor at critical point between real and ideal gas? - Quora

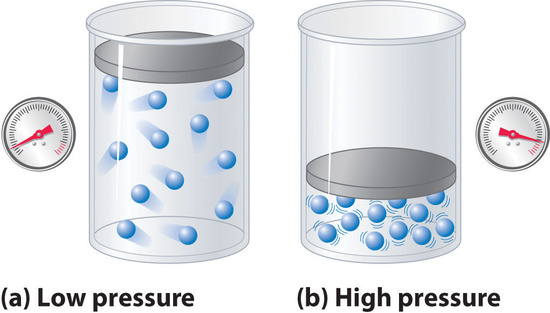
1.gases are compressible 2.gases uniformly expand to fill a container 3. gases form homogeneous mixtures regardless of identity Gases vs. solids & liquids. - ppt download
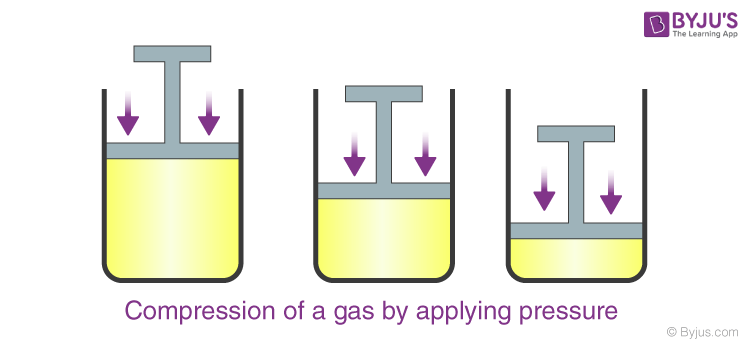
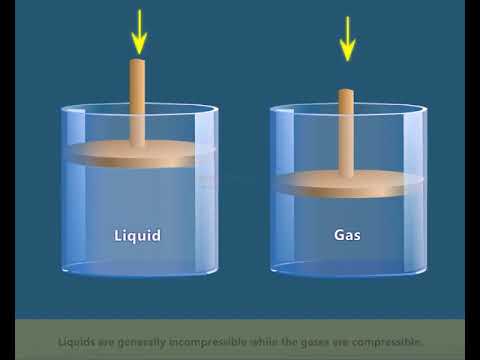
Demonstrate that the gases are highly compressible as compared to solids and liquids. - Sarthaks eConnect | Largest Online Education Community