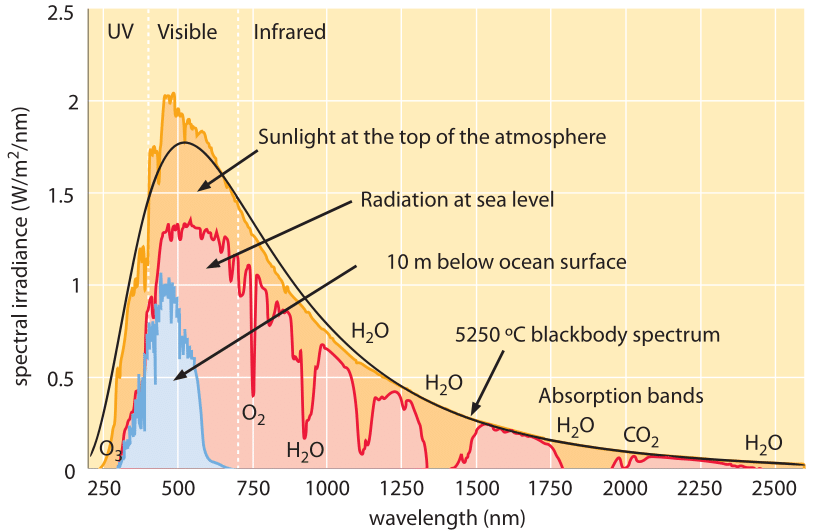
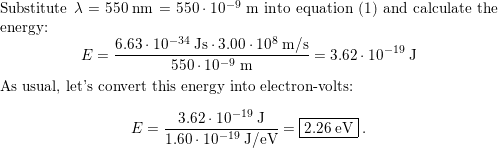Near-field thermophotovoltaics for efficient heat to electricity conversion at high power density | Nature Communications
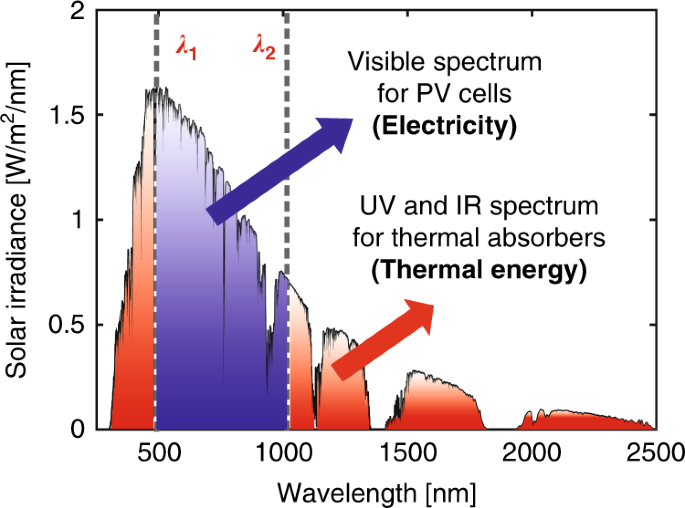
Efficiency limits of concentrating spectral-splitting hybrid photovoltaic-thermal (PV-T) solar collectors and systems | Light: Science & Applications
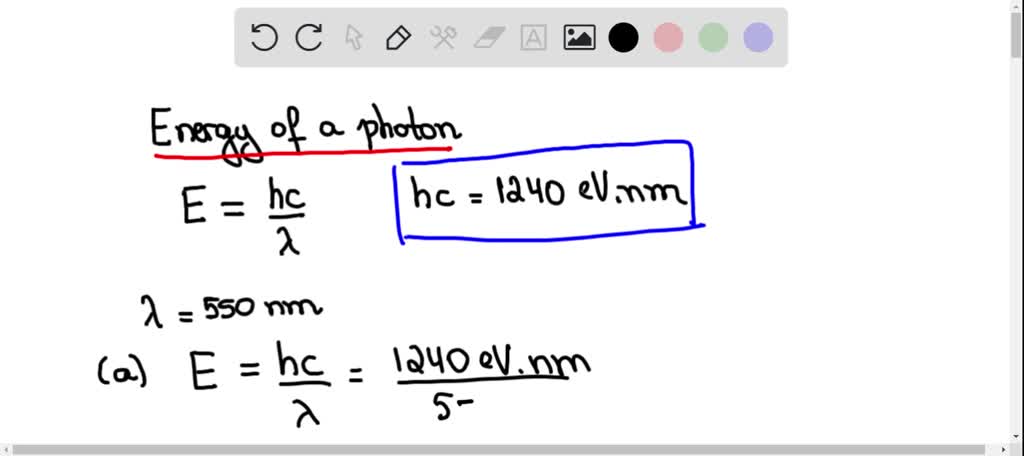
SOLVED:A firefly glows by the direct conversion of chemical energy to light. The light emitted by a firefly has peak intensity at a wavelength of 550 nm a. What is the minimum

Get the Basics Right: Jacobian Conversion of Wavelength and Energy Scales for Quantitative Analysis of Emission Spectra | The Journal of Physical Chemistry Letters

Calculating particle properties of a wave Ch. 12 A light wave consists of particles (photons): The energy E of the particle is calculated from the frequency. - ppt download

Calculating particle properties of a wave Ch. 12 A light wave consists of particles (photons): The energy E of the particle is calculated from the frequency. - ppt download