Glowing Neon Line Sulfur Dioxide SO2 Gas Molecule Icon Isolated on Black Background. Structural Chemical Formula and Stock Vector - Illustration of color, neon: 237105911

Neon gas 99+% in 1" glass vial under normal pressure, element 10 sample : Amazon.de: Business, Industry & Science

Reaction formulas and reaction types in discharge process of neon gas. | Download Scientific Diagram

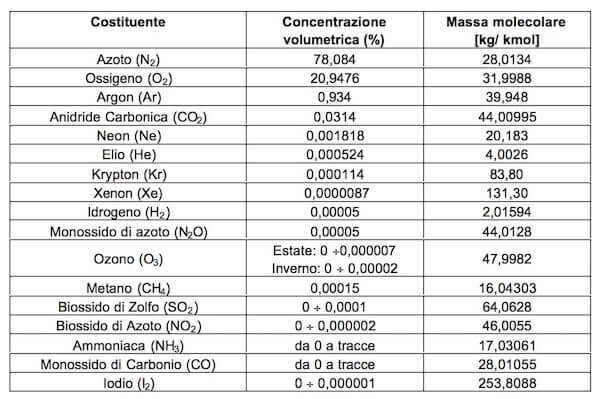
A neon - dioxygen mixture contains 70.6 g a dioxygen and 167.5 g neon. if pressure of the mixture of in the cylinder is 25 bar. what is the partial perssure of
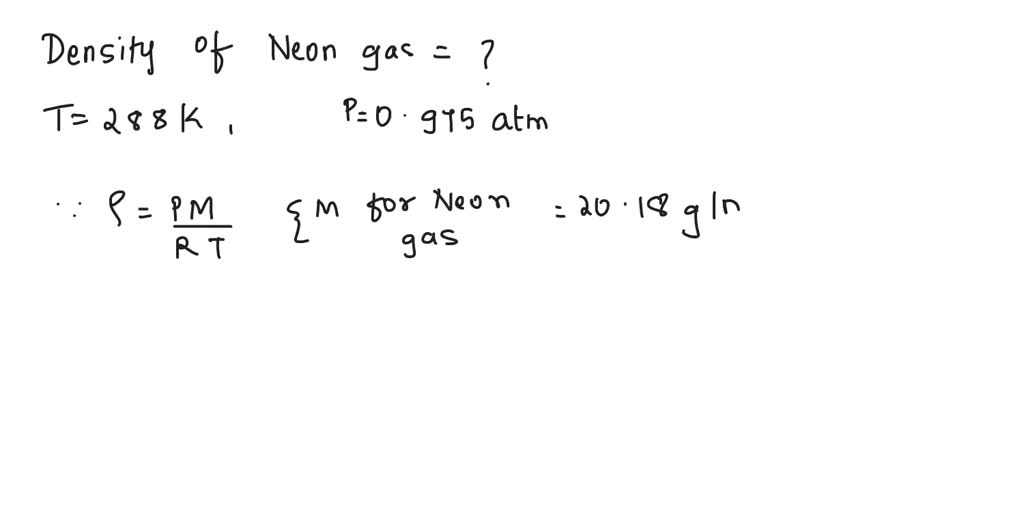
SOLVED: Chapter I Exercise #2: STP = atm and 0 ' What would the new volume of gas be if the gas is changed to STP? The volume of the gas was

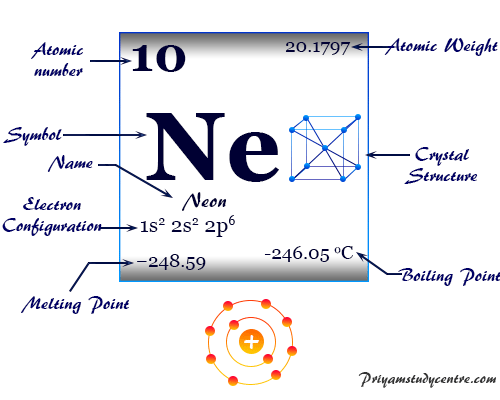
The relative atomic mass of Neon is 20.18 amu. What is mass of 2 moles of Neon gas. A. 40.36 amu B. 40.36g C. 10.09 amu D. 10.09 g | NMDCAT 2022