
Aluminium Nitrate, chromium Nitrate, chromiumii Chloride, ironiii Nitrate, Trigonal planar molecular geometry, molar Mass, carbonate, nitrate, ligand, chromium | Anyrgb
![Ligand steric effect on the coordination mode of nitrite ion: Syntheses and X-ray single crystal structures of trans-[NiL2(ONO)2] and cis-[NiL′2(O2N)](NO2)·H2O [L = 2-(aminomethyl)pyridine and L′ = (2-aminoethyl)pyridine] - ScienceDirect Ligand steric effect on the coordination mode of nitrite ion: Syntheses and X-ray single crystal structures of trans-[NiL2(ONO)2] and cis-[NiL′2(O2N)](NO2)·H2O [L = 2-(aminomethyl)pyridine and L′ = (2-aminoethyl)pyridine] - ScienceDirect](https://ars.els-cdn.com/content/image/1-s2.0-S0022286006008672-gr5.jpg)
Ligand steric effect on the coordination mode of nitrite ion: Syntheses and X-ray single crystal structures of trans-[NiL2(ONO)2] and cis-[NiL′2(O2N)](NO2)·H2O [L = 2-(aminomethyl)pyridine and L′ = (2-aminoethyl)pyridine] - ScienceDirect

The structure of complexes of thorium nitrate with ligand V containing... | Download Scientific Diagram

Synchrotron X-ray-induced photoreduction of ferric myoglobin nitrite crystals gives the ferrous derivative with retention of the O-bonded nitrite ligand. - Abstract - Europe PMC

Ligand Conversion in Nanocrystal Synthesis: The Oxidation of Alkylamines to Fatty Acids by Nitrate | JACS Au

A Comparison of Nitrate Release from Zn/Al-, Mg/Al-, and Mg–Zn/Al Layered Double Hydroxides and Composite Beads: Utilization as Slow-Release Fertilizers | ACS Omega
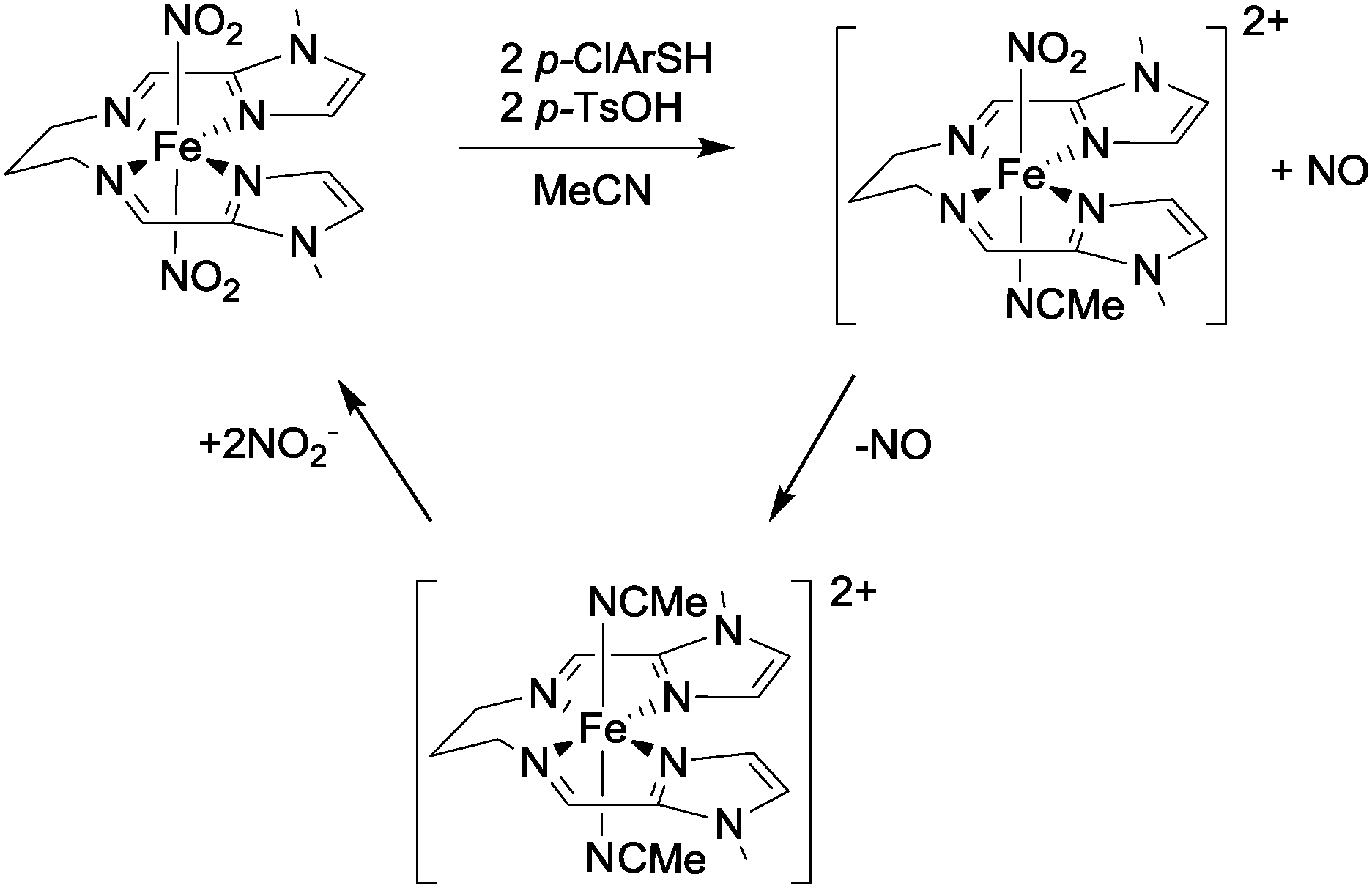
Converting between the oxides of nitrogen using metal–ligand coordination complexes - Chemical Society Reviews (RSC Publishing) DOI:10.1039/C5CS00269A

Figure 1 from The silver(I) nitrate complex of the ligand N-(pyridin-2-ylmethyl)pyrazine-2-carboxamide: a metal–organic framework (MOF) structure | Semantic Scholar
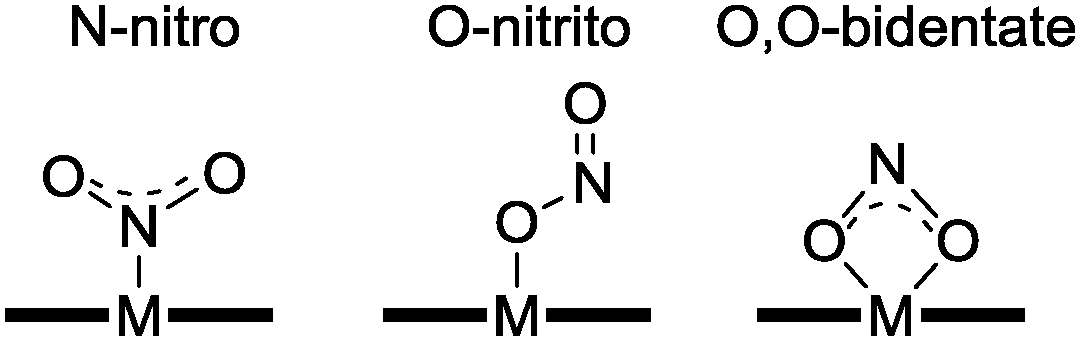
Converting between the oxides of nitrogen using metal–ligand coordination complexes - Chemical Society Reviews (RSC Publishing) DOI:10.1039/C5CS00269A

Ligand effects on nitrate reduction by zero-valent iron: Role of surface complexation - ScienceDirect
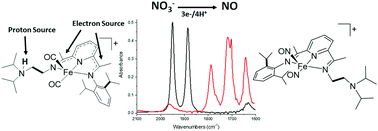
Ligand-based reduction of nitrate to nitric oxide utilizing a proton-responsive secondary coordination sphere - Chemical Communications (RSC Publishing)
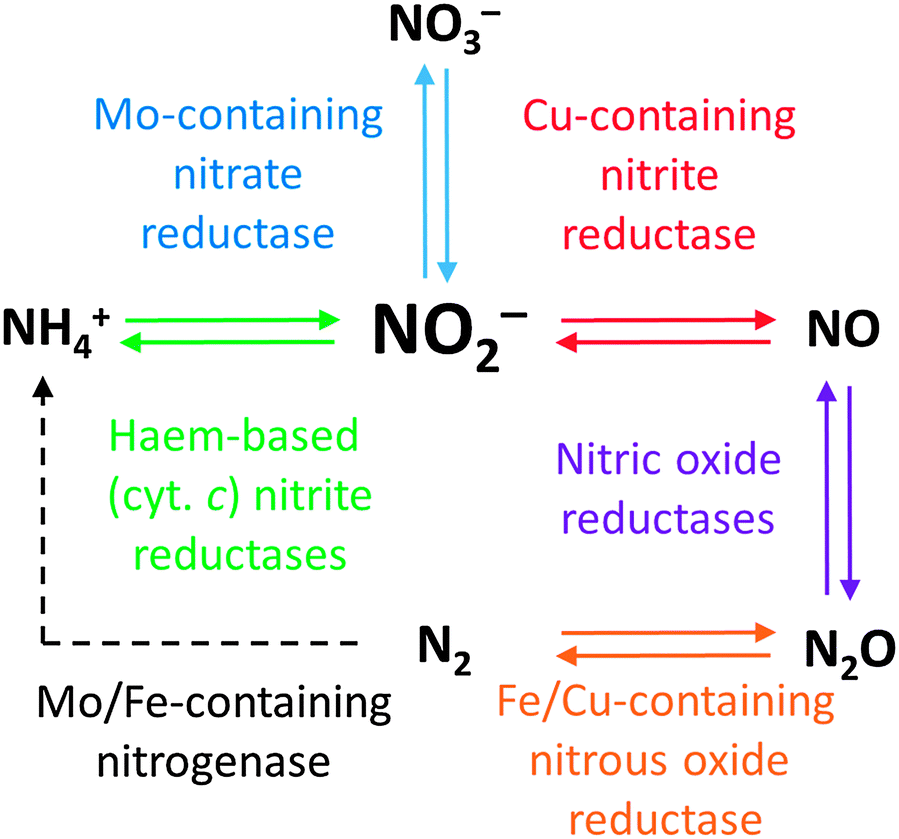
Converting between the oxides of nitrogen using metal–ligand coordination complexes - Chemical Society Reviews (RSC Publishing) DOI:10.1039/C5CS00269A








