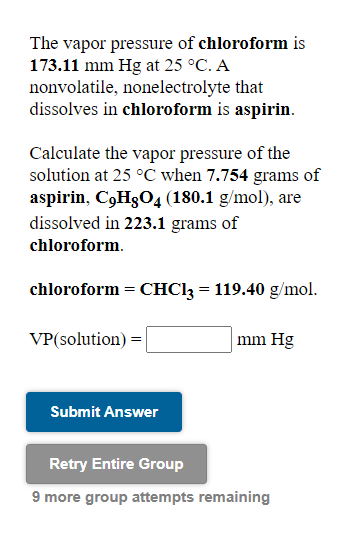
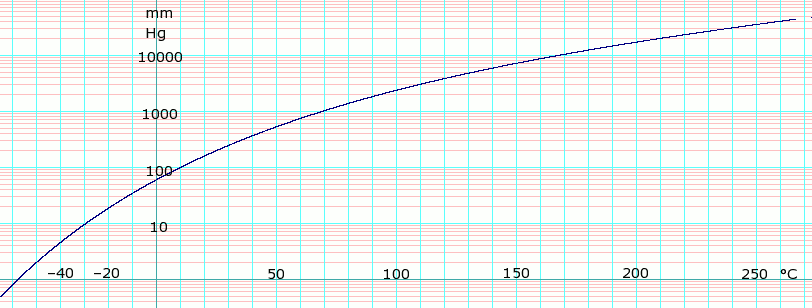
SOLVED: At a certain temperature the vapor pressure of pure chloroform CHCI;) is measured to be 0.74 atm Suppose solution is prepared by mixing 69.2 g of chloroform and 92.7 g of

A solution contains 4.08 g of chloroform (CHCl3) and 9.29 g of acetone (CH3COCH3). The vapor pressures at - brainly.com

SOLVED: The vapor pressure of chloroform (CHCl3) is 55.6 Torr at -7.1 °C. It has an enthalpy of vaporization of 29.2 kJ/mol. Calculate (in °C) chloroform's normal boiling point. Four ice cubes
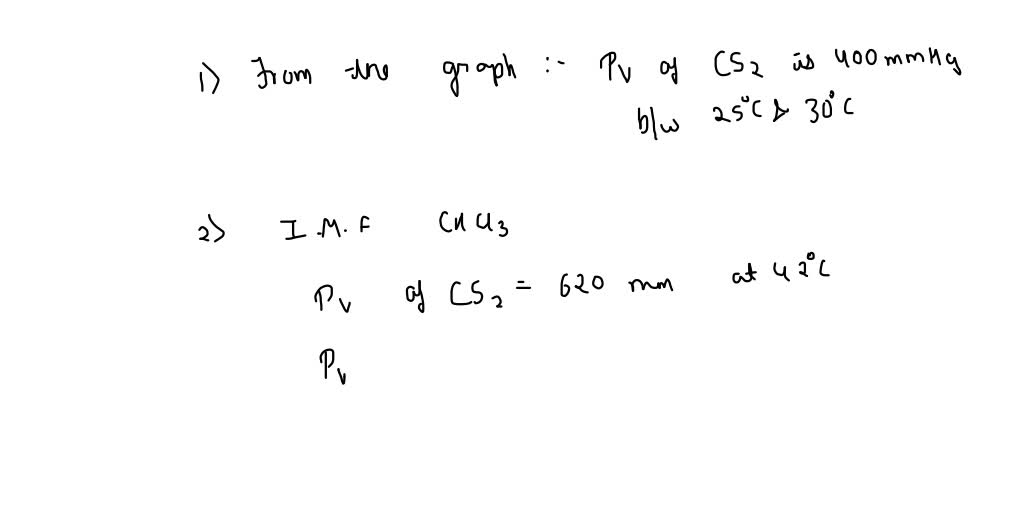
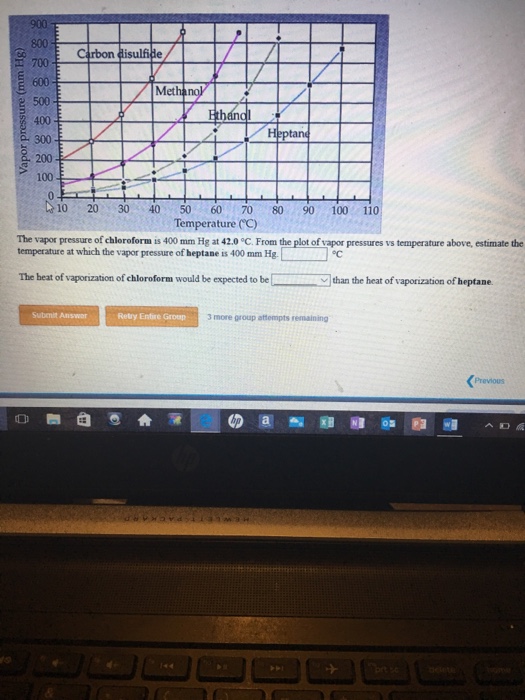
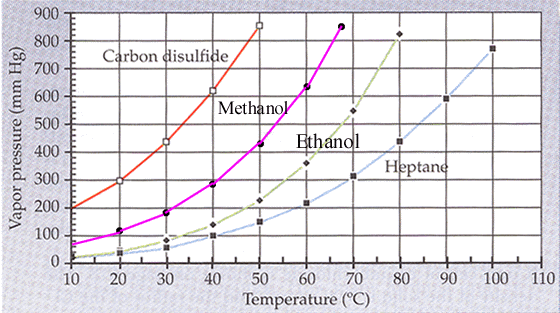
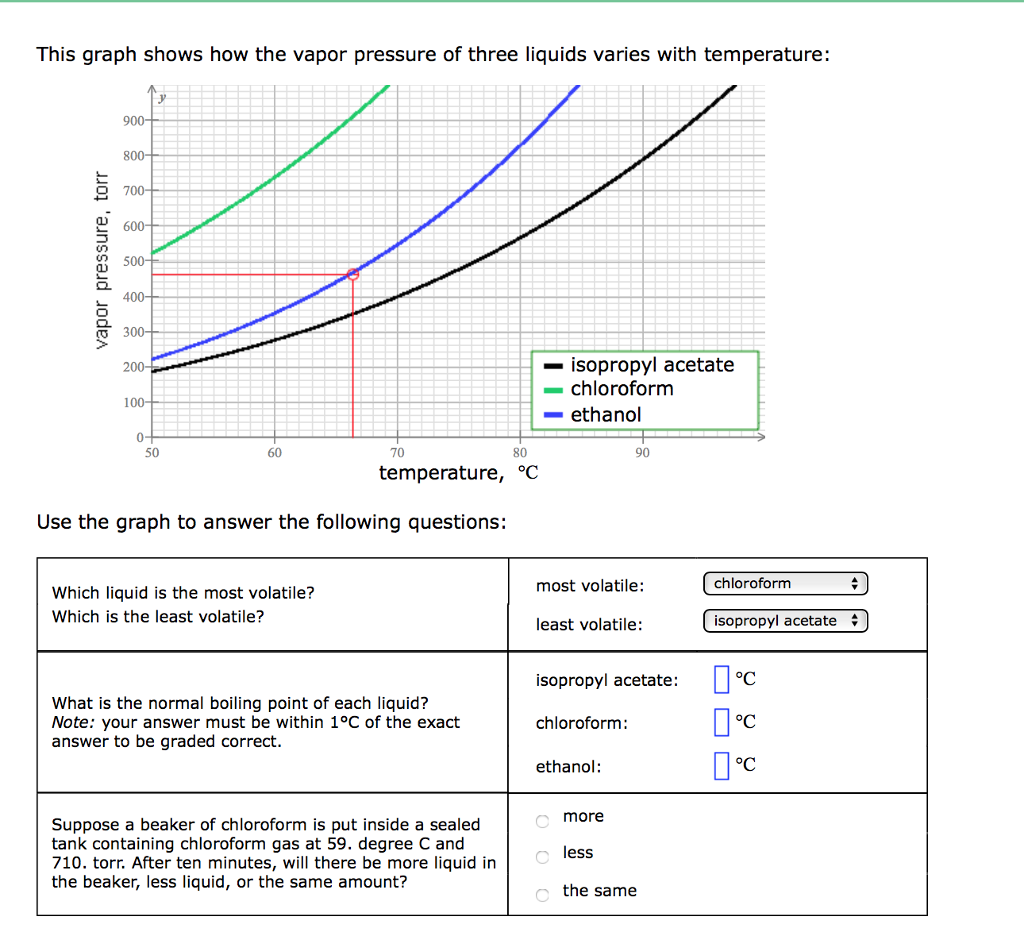
SOLVED: The vapor pressure of chloroform is 400 mm Hg at 42.0 °C. From the plot of vapor pressures vs temperature above, estimate the temperature at which the vapor pressure of ethanol

Vapour pressures of pure acetone and chloroform 328 K are 741.8 mm Hg and 632.8 mm Hg respectively. Assuming that they form ideal solution over the entire range of composition, plot ptotal,pchloroform,

OneClass: Chloroform, CHCl3, has a vapor pressure of 197 mmHg at 23.0 °C, and its heat of vaporizati...

QUESTION 2.37 Vapour pressures of pure acetone and chloroform at 328 K are 741.8 mm Hg and 632.8 mm Hg respectively. Assuming that they form ideal solution over the entire range of

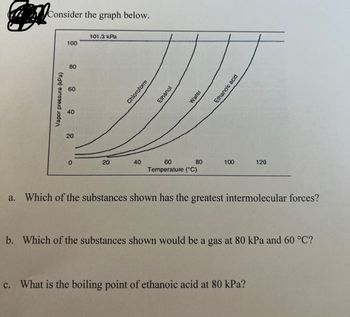
Use the following vapor pressure diagram to estimate the partial pressure of chloroform. | Homework.Study.com

Vapour pressure of chloroform (CHCl3) and dichloromethane (CH2Cl2) at 298K are 200mm Hg and 415mm Hg - YouTube
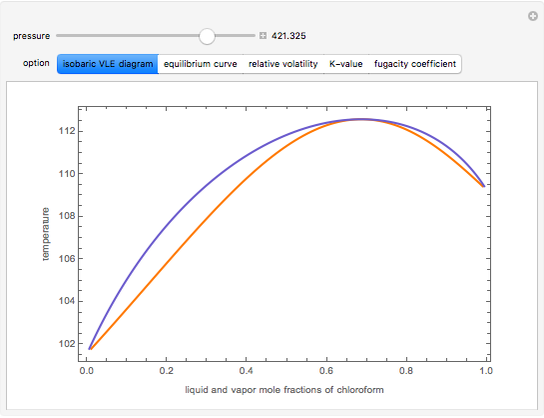
High Pressure Vapor-Liquid Equilibrium Data of a Binary Mixture of Chloroform and Acetone - Wolfram Demonstrations Project

To what temperature should water be heated so that its vapor pressure equals the vapor pressure chloroform 40oC?

SOLVED: 13 Using the data below, calculate the vapor pressure of chloroform over a chloroform-benzene solution at 25°C, which contains 50.0 g of CHCl3 and 50.0 g of C6H6. Assume that the